IBCSG 67-22 / PREcoopERA
EUCT-No: 2022-503013-32
Sponsor: ETOP IBCSG Partners Foundation
Introduction / Overview
A window-of-opportunity study comparing giredestrant +/- triptorelin versus anastrozole + triptorelin in premenopausal patients with early-stage ER-positive/HER2-negative breast cancer.
This study investigated new hormonal treatment options for premenopausal women with early-stage, ER-positive/HER2-negative breast cancer. During the short period before surgery, the new drug giredestrant was tested to see how well it can slow tumor growth. To this end, the sponsor is comparing giredestrant—either alone or in combination with triptorelin—with the current standard treatment of anastrozole and triptorelin. The goal is to determine whether giredestrant is more effective within four weeks or whether it works just as well without additional suppression of ovarian function. The sponsor aims to help make future hormonal therapies for younger patients even more effective and, at the same time, gentler.
News
Participation in the study was successfully completed in July 2025. Data analysis was finalized at the end of 2025, and the study has since been officially concluded. The results will be presented at the ESMO Breast Congress, and a scientific paper has already been submitted. Further information will follow as soon as it is published.
Design
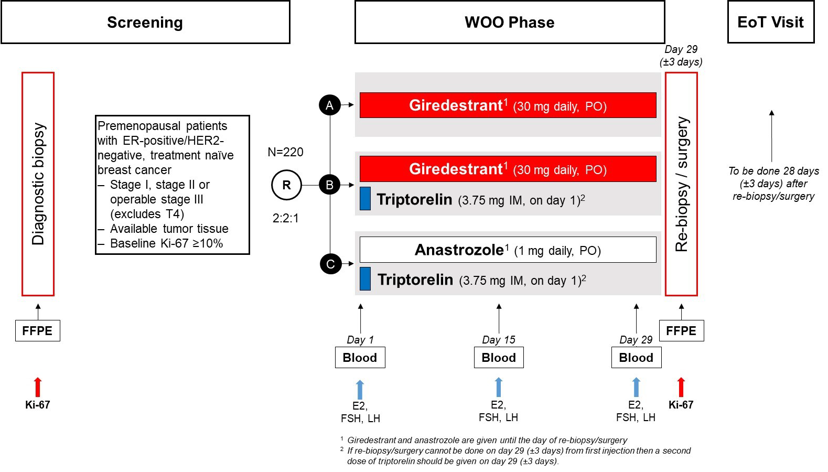
Randomized (2:2:1), multicenter, open-label, three-arm (A, B, C), Window-of-Opportunity (WOO) trial to evaluate the activity and safety of giredestrant (A) versus giredestrant plus triptorelin (B) versus anastrozole plus triptorelin (C).
Study Design Precoopera