Introduction
A Study of Camizestrant in ER+/HER2- Early Breast Cancer After at Least 2 Years of Standard Adjuvant Endocrine-Based Therapy.
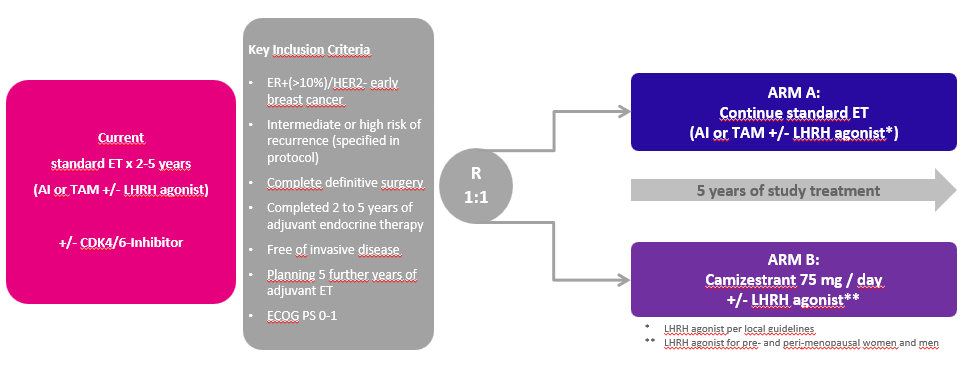
The target population of interest in this study consists of male and female patients with ER+/HER2- early breast cancer, who have completed definitive locoregional therapy and at least 2 years of standard adjuvant ET, with or without a cyclin dependent kinase 4 and 6 (CDK4/6) inhibitor, and who are currently disease-free but with intermediate or high risk of recurrence.
News
Details about the study, inclusion and exclusion criteria and participating trial sites can be found on ClinicalTrials.gov.
We are still looking for patients for this study.
You are welcome to refer a patient to one of our trial sites. An overview of all participating study sites can be found at the end of this page or on the Reesi platform.
Here you will find the link to the login area of Reesi.

Design
Primary Efficacy
Comparison of IBCFS (invasive breast cancer-free survival) rates by treatment
Patient Profile
- Early ER+/HER2- breast cancer with no disease recurrence
- Pre-, Peri-, and postmenopausal Women and Men
- Adequate surgical and systemic pre-therapy with or without chemotherapy or radiotherapy
- At least 2 years and up to 5 years of standard adjuvant ET
- Patients may have received adjuvant CDK4/6 inhibitors prior to study entry
- Intermediate or high risk of recurrence as defined per protocol
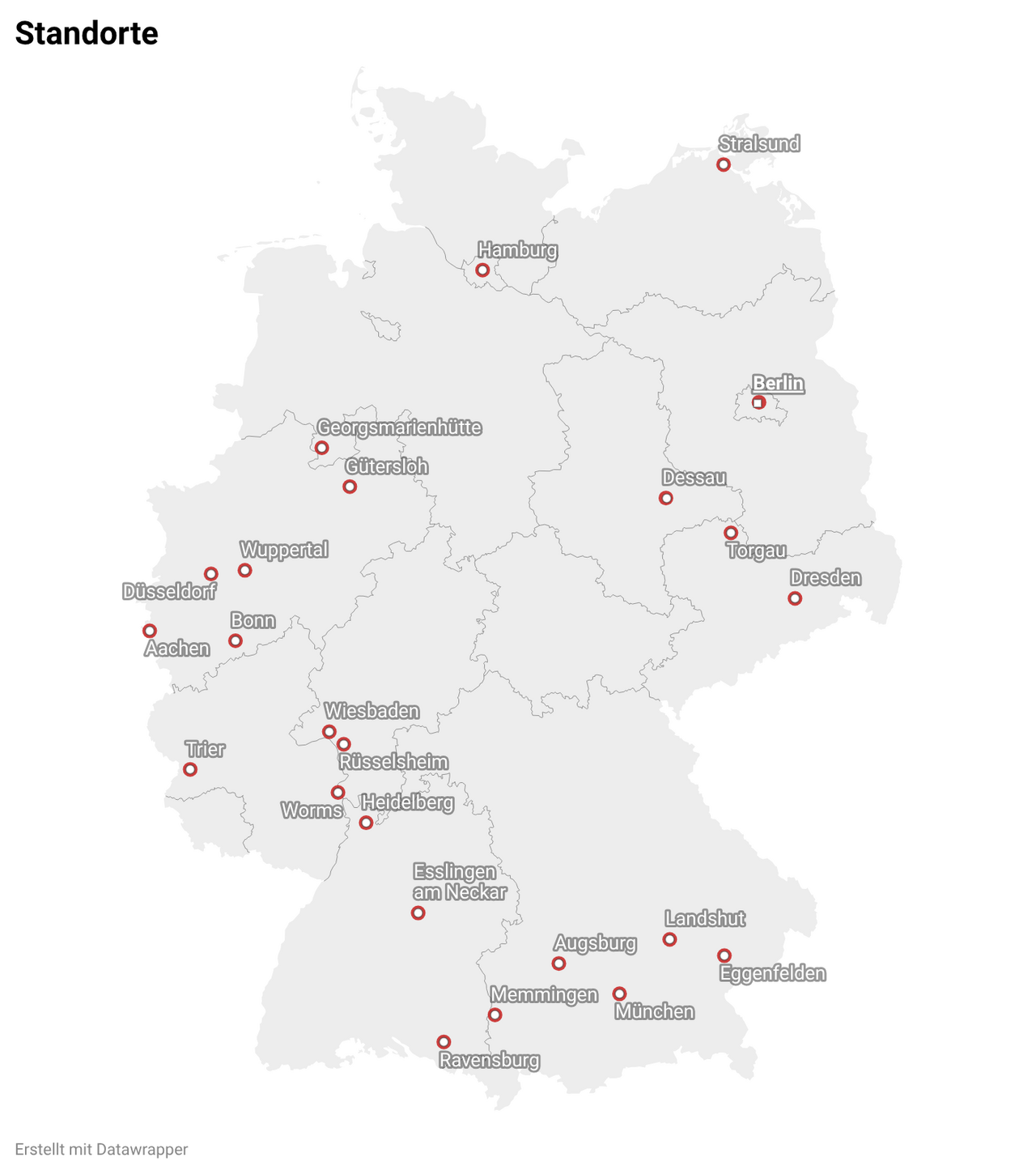
The CAMBRIA-1 study is currently (as of April 2026) being conducted at 27 study sites in Germany:
- Aachen Universitätsklinikum Aachen, Prof. Dr. Elmar Stickeler
- Augsburg Hämatologie-Onkologie im Zentrum MVZ GmbH, Dr. Bernhard Heinrich
- Berlin DBZ Onkologie GmbH, Dr. Antje Müller
- Bonn Gynäkologisches Zentrum Bonn, PD Dr. Christian Kurbacher
- Dessau-Roßlau Städtisches Klinikum Dessau, Dr. Hermann Voß
- Dresden Gemeinschaftspraxis Hämatologie Onkologie, PD Dr. Thomas Illmer
- Dresden Universitätsklinikum Carl Gustav Carus an der TU Dresden, Dr. Theresa Link
- Düsseldorf MVZ Medical Center / GynOnco, Dr. Athina Kostara
- Düsseldorf Universitätsklinikum Düsseldorf, Prof. Dr. Tanja Fehm
- Eggenfelden Studienzentrale für das MVZ Eggenfelden e.K., Dr. Jürgen Terhaag
- Esslingen Klinikum Esslingen GmbH, PD Dr. Alexander Hein
- Georgsmarienhütte Niels Stensen Kliniken / MVZ II & Senologie, Dr. Kerstin Lüdtke-Heckenkamp
- Gütersloh Onkodok GmbH, Dr. Reinhard Depenbusch
- Hamburg Gynäkologische Praxisklinik Hamburg-Harburg, Dr. Christoph Großmann
- Hamburg Überörtliche Gemeinschaftspraxis Dres. Wierecky & Brandl, Dr. Elke Hennes
- Heidelberg Nationales Centrum für Tumorerkrankungen (NCT), Dr. Laura Michel
- Landshut VK&K Studien GbR/ÜBAG MVZ Dr. Vehling-Kaiser GmbH, PD Dr. Lorenz Rieger
- Memmingen Klinikum Memmingen, PD Dr. Felix Flock
- München Rotkreuzklinikum München, Dr. Claus Hanusch
- Ravensburg Studienzentrum Onkologie Ravensburg GbR, Prof. Dr. Thomas Decker
- Rüsselsheim GPR Klinikum Rüsselsheim, Dr. Adriana Haus
- Stralsund Gynäkologie Kompetenzzentrum, Dr. Carsten Hielscher
- Torgau Kreiskrankenhaus Torgau Johann Kentmann, Dr. Eike Simon
- Trier Klinikum Mutterhaus der Borromäerinnen, Prof Dr. Sebastian Jud
- Wiesbaden HELIOS Dr. Horst Schmidt Kliniken, Prof. Dr. Michael Eichbaum
- Worms Klinikum Worms, Dr. Sebastian Züfle
- Wuppertal HELIOS Universitätsklinikum Wuppertal, Prof. Dr. Vesna Bjelic-Radisic
Contact
Anna Huber