Our Services for Your Success
You are planning your next trial? - You found your spot!
From study design over delivery to final report, we support you with scientific expertise, clinical experience, and a strong international network.
GBG - Your Partner in Therapeutic Development
Academic Excellence
Over 20 years of experience in breast cancer research and in-depth scientific expertise.
Clinical Implementation
End-to-end services for Phase Ib to IV trials—nationally and globally.
Unique Resources
Access to a large network as well as comprehensive biobank and cryobank data.
Full-service clinical trial support
- Feasibility
- Regulatory & Start-up
- Project Management & Monitoring
- Data Management & Biostatistics
- Medical Writing & Pharmacovigilance
- Quality Management
Specialized GBG Services
- PRO-based Long-Term Follow-Up
- Central Pathology & Quality Control
- Biomaterial Banking & Translational Research
- Sponsorship & Study Design (IITs)
- Workshops & Scientific Consulting

Our Services in Detail
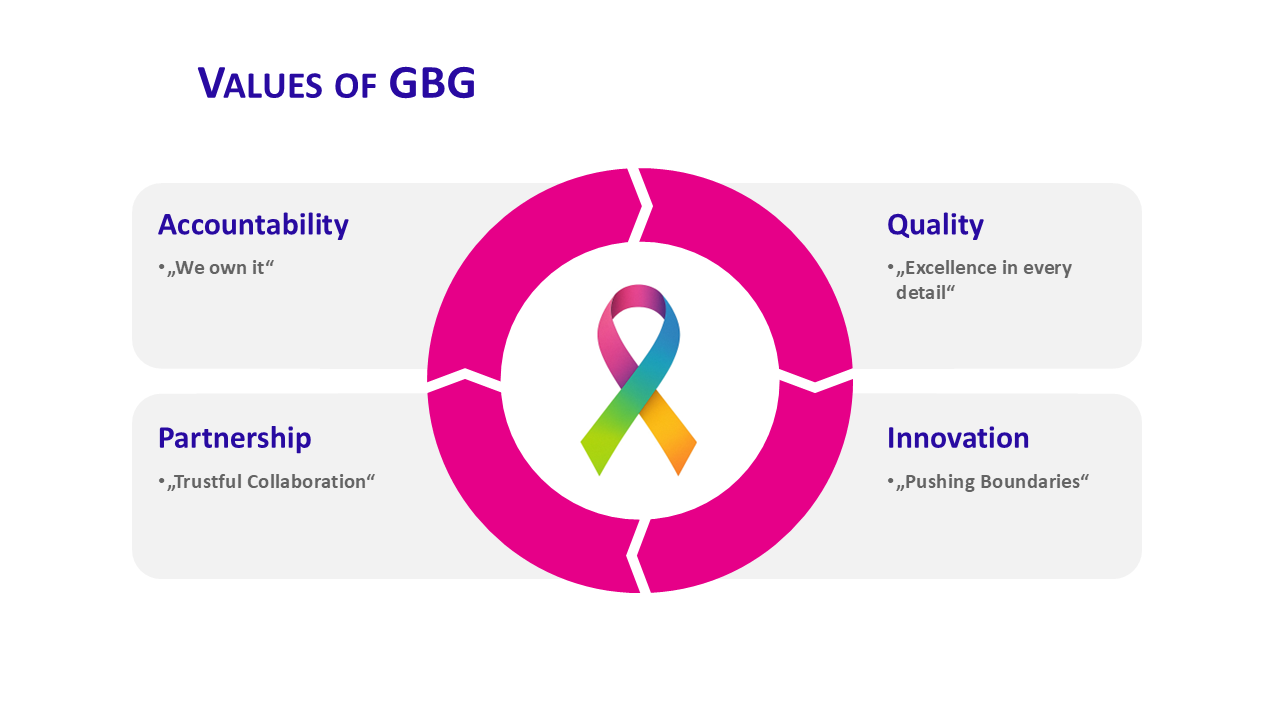
Accountability
Full responsibility for our scientific and clinical research projects
Ensuring the careful, reliable, and effective implementation of every project
Partnership
Flexible partnership models with real added value
Accelerating the path from research to effective cancer therapies
Quality
Implementation in accordance with the highest internal and external standards
Committed to results that advance oncological research
Innovation
Openness to new approaches and innovative technologies
Improving treatment outcomes for patients worldwide
Our Services in Detail
- Preparation of study protocols and all associated documents (e.g., informed consent forms)
- Conception and development of study concepts and protocols for investigator-initiated studies in which GBG acts as the sponsor
- Medical monitoring of clinical data (e.g., laboratory values, identification of outliers)
- Preparation of reports on SUSARs and/or SAEs
- Pharmacovigilance activities (review and evaluation of SAEs and SUSARs, preparation of DSURs)
- Preparation of clinical study reports (CSRs)
- Evaluation of reference documents (e.g., Investigator’s Brochure)
- Participation in the planning and execution of data analyses
- Support with medical coding (e.g., WHO, MedDRA, ICD)
- Active preparation for and participation in sub-board meetings with the GBG KOL network
- Organization of workshops for investigators (e.g., presentation of new projects, active ingredients, management of adverse events)
+ Additional service:
- Submissions to the EMA and ClinicalTrials.gov upon study completion
- Publication of clinical studies in renowned journals through:
- Coordination of the annual publication strategy
- Presentations at international conferences
- Publication of manuscripts in renowned international journals
- Communication with numerous international stakeholders (authors, collaborating partners, and pharmaceutical partners)
- Strong Project Management Team
- A large team of project managers with experience in oncology research supports projects in all phases, from Phase I to Phase IV.
- Close Relationships with Study Sites
- Thanks to GBG’s close relationships with study sites, our project managers ensure a quick and smooth start to drive and secure patient recruitment.
- Central Point of Contact for the Study
- Coordination of internal teams to ensure successful execution and seamless communication among all stakeholders.
- Global Coverage
- GBG’s core project management team is based in Germany. Thanks to strong global partnerships, we can successfully manage large Phase Ib through IV studies.
- Partnership-oriented approach
- Our teams act as partners to our sponsors and support study conduct on an equal footing.
- Operational backbone
- Full operational oversight throughout the entire project. Proactive risk management from the outset to ensure smooth study progression.
- Close collaboration with GBGs study sites – selecting the right sites from the start
- CDAs and MSAs signed with the majority of study sites
- Utilization of fast-track sites to achieve a turnaround time of 2–6 weeks for document collection and contract signing with the sites
- Experienced regulatory team to support submissions in EU and non-EU countries
- Close collaboration with international study groups to shorten start-up times
- Excellent project management team to support sites
Finding the right center for your project
- Extensive database of investigators and study centers
- Existing CDAs and MSAs with many of our GBG network centers
- Close collaboration through subcommittees, workshops, ASM, etc.
- Clear understanding of the situation at the study sites
Selection of suitable sites from the outset to minimize the number of sites without recruitment
- Careful review of feasibility results
- Sites with Fast-Track status / Priority 1
- Existing MSA + rapid ramp-up + good recruiters + no PSSV required + suitable patient pool
- Priority 2 sites
- Existing relationships / MSA + good recruiters + suitable patient pool
- Final decision in close collaboration with the sponsor
Regulatory expertise
- Submission to ethics committees and authorities (BfArM & PEI)
- Voluntary Harmonization Procedure (VHP)
- CTIS (Part I and Part II)
- IVDR
- MDR
- International experience outside the EU
Ensuring high data quality from clinical trial sites
- Strong monitoring team with scientific and medical backgrounds
- Support for on-site and remote monitoring visits
- Coverage from Phase Ib to IV
- Risk-based monitoring
- Targeted and event-driven monitoring plans
- Close relationships with investigators at study sites
National and international experience – support for small to large pharmaceutical and biotech companies
Standard services:
- Coverage of all aspects of clinical monitoring visits
- Experience in drug accountability in open-label and blinded studies, regardless of the route of administration
Additional services:
- Support for sponsor audits
- Support for regulatory inspections
- Close collaboration with participating hospital pharmacies
- Up-to-date in accordance with ICH GCP E6/R3
- Protocol review
- Patient profiles
- CRF design
- Setup and maintenance of the EDC system
- Edit checks/data validation
- User management
- Data entry guidelines
- Training
- Data cleansing/queries
- SAE/AE reconciliation
- Reporting
- Documentation overviews
- Department-specific tools
- Central database
- Archiving
- Statistical design of clinical trials (sample size, endpoints, alpha level, randomization, stratification)
- Interim and final analyses to ensure scientific validity
- Support for independent data monitoring committees
- PRO/QoL analyses
- Support for publications and regulatory submissions
- Contribution to translational research through biostatistical modeling
- PK/PD analyses
- Pooled and meta-analyses
- Bayesian methodology
- Systems: SAS, R (analysis), EAST, nQuery Advisor (sample size)
- Preparation and review of protocols in accordance with international standards
- Preparation of study reports
- Support with manuscripts and publications
- Posters
- Organization of workshops
- Conference presentations
- Literature review and knowledge translation
- Training and continuing education materials
- Controlled Documents
- Development and Monitoring of Training Programs
- Quality Management System
- Supplier Qualification
- Audits and Inspections
- Management of Corrective and Preventive Actions (CAPA)
- Support for the Validation of Internal Systems
- Risk Management
- Point of Contact for All Questions Regarding Quality Management
- Management Report
- Internal and External Inspections
- Extensive biobank and cryobank resources for biomarker projects
- Sample tracking and data management, data reconciliation
- Labkit management, assembly, and distribution
- Logistics for biosamples, nationally and internationally
- On-site support and training, sample management guidelines, laboratory manual
- Reporting and communication, sample status reports, planning of biomarker analysis
- Coordination of translational research projects, processing of sample requests
- Compliance with legal and ethical regulations regarding the collection of biological samples
Long-term storage of various biomaterials from clinical trials:
- Whole blood
- Plasma
- Serum
- Urine
- Feces
- Fresh-frozen tissue
- DTCs from bone marrow (disseminated tumor cells)

Contact
Senior Director Business Development